 |
 |
- Search
| Genomics Inform > Volume 19(2); 2021 > Article |
|
Abstract
Breast cancer is one of the leading causes of cancer in women all over the world and accounts for ~25% of newly observed cancers in women. Epigenetic modifications influence differential expression of genes through non-coding RNA and play a crucial role in cancer regulation. In the present study, epigenetic regulation of gene expression by in-silico analysis of histone modifications using chromatin immunoprecipitation sequencing (ChIP-Seq) has been carried out. Histone modification data of H3K4me3 from one normal-like and four breast cancer cell lines were used to predict miRNA expression at the promoter level. Predicted miRNA promoters (based on ChIP-Seq) were used as a probe to identify gene targets. Five triple-negative breast cancer (TNBC)ŌĆÆspecific miRNAs (miR153-1, miR4767, miR4487, miR6720, and miR-LET7I) were identified and corresponding 13 gene targets were predicted. Eight miRNA promoter peaks were predicted to be differentially expressed in at least three breast cancer cell lines (miR4512, miR6791, miR330, miR3180-3, miR6080, miR5787, miR6733, and miR3613). A total of 44 gene targets were identified based on the 3ŌĆ▓-untranslated regions of downregulated mRNA genes that contain putative binding targets to these eight miRNAs. These include 17 and 15 genes in luminal-A type and TNBC respectively, that have been reported to be associated with breast cancer regulation. Of the remaining 12 genes, seven (A4GALT, C2ORF74, HRCT1, ZC4H2, ZNF512, ZNF655, and ZNF608) show similar relative expression profiles in large patient samples and other breast cancer cell lines thereby giving insight into predicted role of H3K4me3 mediated gene regulation via the miRNA-mRNA axis.
Breast cancer is one of the leading causes of death in women all over the world [1]. There are many subtypes in breast cancer identified based on the origin, hormone receptors expression, and response to treatment. There are four basic subtypes namely luminal-A, luminal-B, human epidermal growth factor receptor-2 (HER2) and triple-negative breast cancer (TNBC) [2]. Among all the subtypes, the TNBC is very aggressive and has poor prognosis compared to other subtypes and very few systemic treatment options are available other than chemotherapy [3]. Luminal-A (estrogen receptor [ER]+ and/or progesterone receptor [PR]+, HER2ŌłÆ) because of ER expression, has better prognosis compared to TNBC [2]. Epigenetic regulation via histone modification of microRNA (miRNA) promoters is known to play a crucial role in breast cancer regulation [4]. Histone modifications wrapped around genes play an important role in gene regulation by providing access to transcription factors, RNA-polymerases, and other regulatory mechanisms [5]. There are several histone post-translational modifications identified earlier with a unique regulatory function for each of them. Using chromatin immunoprecipitation followed by sequencing (ChIP-Seq), one can identify the position of targeted protein binding (transcription factors, histone modifications) regions in the genome [6].
Epigenetic gene regulation occurs in three major ways namely DNA methylation of CpG islands, histone modifications, and non-coding RNA mediated [7]. Each regulatory level has a crucial role in normal cell development and diseases such as cancer and other non-communicable diseases. Gene level histone modifications can be used to predict the status of a gene whether it is active or inactive. H3K4me3 modification at the promoter level provides information about active genes, whereas H3K27me3 and H3K9me3 modifications provide the inactive status of the gene [8]. Previous studies provided evidence for correlation of histone modifications (H3K36me3, H3K9ac, H3K27ac, and H3K4me1) with gene expression. Combinations of two or more histone modifications at gene promoter and gene body level provides more resolution to predict gene activity [8].
Non-coding (nc) RNAs (LncRNAs, long intervening/intergenic noncoding RNA, miRNA, and small interfering RNA) play a major role in gene regulation of different biological processes such as cell cycle and proliferation along with developmental and metabolic processes [9]. The most widely studied ncRNAs are miRNAs which are small ncRNA ~22nt in length, evolutionarily conserved, and have a wide regulatory role in development and diseases. They play an important role in gene regulation by targeting complementary binding sites in untranslated region (UTR) regions of gene transcripts or by targeting promoters or other miRNA or LncRNA [10]. Interestingly, miRNAs are also involved in the upregulation of specific genes via binding to their promoters in the nucleus and thereby controlling gene expression [11]. An actively transcribed miRNA is able to regulate ~100ŌĆÆ1,000 genes by complementary binding to their targeted genes. Based on conserved base-pairing homology, it has been predicted that ~60% of human genes are targeted by miRNAs [12]. In many cancers dysregulation of miRNA causes cancer progression and drug resistance. The role of miRNA deregulation in breast cancer was first reported in 2005 wherein the role of miR548 as an oncogenic regulator in breast cancer was elaborated [13]. There are other miRNAs such as let-7, miR145, miR200, and miR497 with a definitive role in breast cancer [14].
The present study is limited to normal-like, luminal-A and TNBC-claudin subtypes of breast cancer based on hormonal receptor expression. One each of normal subtype, ER positive subtype (Luminal-A) and ER negative (TNBC-claudin) were chosen for analysis. In the current study, ChIP-Seq data corresponding to histone modification H3K4me3 for one normal-like (MCF10A) and four breast cancer cell lines (luminal-A [MCF7, ZR751], TNBC [MB231, MB436]) were chosen to understand role of miRNA-gene promoter regulation of miRNA-mRNA axis. An attempt has been made to map the epigenetic expression patterns (histone H3K4me3) with RNA-sequencing (RNA-Seq) expression of miRNA targeted genes. Analysis of such combined data promises to provide insights into understanding epigenetic gene regulation (chromatin) as well as gene expression [15].
ChIP-Seq data were downloaded from Gene Expression Omnibus (GEO) for breast cancer pertaining to six cell lines, viz., normal-like (MCF10A and 76NF2V), luminal-A subtype (MCF7 and ZR751), and TNBC subtype (MB231 and MB436), each with one activation histone modification H3K4me3 and two replicates (sequenced as Illumina single-end reads) (Supplementary Tables 1, 2) [16]. RNA-Seq data for the above-mentioned cell lines with four replicates were also downloaded from GEO (Supplementary Table 3) [16].
The raw reads were checked for quality using FastQC (version 0.11.7) [17]. BWA-MEM (Burrows-Wheeler alignment-Maximal Exact Matches) version 0.7.17 was used for alignment with reference genome build hg38 [18]. Samtools (version 1.8) was used to manage replicates and for sam to bam conversion [19]. ChIP-Seq analysis was done using model-based analysis of ChIP-Seq (MACS2, version 2.1.1.20160309) [20]. Peak calling was done using narrowpeak as H3K4me3 generates narrow histone marks [21]. p-value thresholds for peak calling were set to 0.001 for all samples and all replicates (Fig. 1A) [22]. Raw ChIP-Seq data of both H3K4me3 and corresponding input sequence was used for peak calling. To identify the reproducibility within the biological replicates IDR2.0.3 tool was used with a threshold of 0.05 to obtain statistically significant peaks [23,24]. Pseudo replicate analysis was carried out to identify low reproducible replicates which satisfy the criteria of N1/N2 Ōēź 2 and Np/Nt Ōēź 2 (where N1 represents the number of replicate1 self-consistent peaks, and N2 represents the number of replicate two self-consistent peaks; Np represents the number of peaks consistent between pooled pseudoreplicates, and, Nt represents the number of peaks consistent between true replicates) [24]. Peaks were annotated using HOMER (v3.12) tool [25]. Peaks corresponding to miRNA promoter genes were extracted using in-house scripts. Cis-regulatory Element Annotation System (CEAS -0.9.9.7) was used to get statistics on ChIP enrichment for genomic features such as chromosomes, promoters, gene bodies, or exons, to infer genes that are most likely to be regulated by a binding factor [26]. For RNA-Seq analysis HISAT2 (v2.1.0) was used for alignment of raw reads [27] and feature counts (v1.5.0p1) tool was used to count reads mapped on to each gene [28]. DEseq2 (v1.24) was used for differential gene expression of subtypes [29]. Downregulated and upregulated genes were selected based on log2fold change > 2 as per guidelines for analysis of multi-omics data [30]. Differential expression of genes for normal-like (MCF10A) vs. luminal-A (MCF7 and ZR751) and normal-like (MCF10A) vs. TNBC (MB231 and MB436) was carried out (Supplementary Fig. 1). miRNA sequences were extracted and checked for complementarity with 3ŌĆ▓-UTRs of downregulated genes. RNAhybrid server (https://bibiserv.cebitec.uni-bielefeld.de/rnahybrid) was used to predict miRNA-mRNA interactions with seedmatch (2ŌĆÆ8 bp) using the helix constraint ŌĆ£fromŌĆØ and ŌĆ£toŌĆØ parameter along with binding energy cutoff of ŌēżŌĆÆ25 kcal/mol [31]. In the current analysis, we used stringent minimum free energy (ŌēżŌĆÆ25 kcal/mol, except for miR3613 (ŌēżŌĆÆ18 kcal/mol) due to low GC content) to predict strong putative targets with high miRNA-mRNA duplex binding stability [31]. These energy cutoffs were used based on previous studies of miR1306-ADAM10 duplex that have been experimentally validated [32]. The workflow for ChIP-Seq and RNA-Seq data integration is depicted in Fig. 1B.
Relative expressions of the gene targets identified were verified using the UALCAN (http://ualcan.path.uab.edu) and CCLE (Broad Institute Cancer Cell Line Encyclopedia, https://portals.broadinstitute.org/ccle) databases. ACTB gene was used as a control for relative expression analysis using the CCLE database. The UALCAN hosts the relative expression of genes across normal versus different cancer types from the TCGA (The Cancer Genome Atlas) cancer resource associated with clinicopathological data [33]. The breast cancer cell line data (60) available in CCLE database was also incorporated into the study for validation. Kaplan-Meier (KM) plots from Human Protein Atlas were used for survival analysis (https://www.proteinatlas.org/).
All the ChIP-Seq datasets passed the quality check (Supplementary Fig. 2) and >86% of reads were mapped to the reference genome for all replicates of H3K4me3 (for each cell line) used in the study. The number of peaks in the biological replicates varied from 27875 to 64652 for different cell lines (Fig. 2). Reproducibility analysis of peaks (obtained for the replicates) enabled identification of statistically significant peaks (threshold 0.05) (Table 1, Supplementary Figs. 3, 4) which are common between biological replicates [21]. In the normal cell line (MCF10A) and luminal-A subtype (cell lines MCF7 and ZR751), 16,601 peaks (59.7%), 13,008 peaks (53.4%) and 10,158 peaks (48.1%) passed the reproducibility threshold respectively. In the triple-negative subtype (cell lines MB231 and MB436), 14,339 peaks (65.7%) and 16,549 peaks (61.2%) passed the threshold. The highest percentage of overlapping peaks between the replicates was observed in cell line MB231 (65.7%) whereas the least percentage of peaks that passed the threshold was seen in cell line ZR751 (48.1%). All cell lines except 76NF2V cell line generated reproducibility Ōēź 2, which indicated that the peaks were reproducible and statistically significant. Hence, cell line 76NF2V was not used for further analysis because of the low reproducibility of replicates, N1/N2 was 3.370 (Supplementary Table 4). Chromosomal-level distribution of ChIP-peaks is available in Supplementary Fig. 5.
miRNA promoter regions were identified for each cell-line (Table 2, Supplementary Table 5). Peaks corresponding to miRNA-gene promoters that are common and unique between normal versus cancerous cell lines were identified (normal vs. TNBC, normal vs. luminal-A, and TNBC vs. luminal-A) (Fig. 3, Tables 3,4). The majority of the miRNAs predicted have been reported to have a role in breast cancer (Supplementary Table 6).
Cell lineŌĆÆspecific miRNAs obtained in this study have been listed in Table 3. Few of these miRNAs have been validated previously [34]. It is interesting to note that there are no common miRNAs between both the luminal-A cell lines used in this study; however, five TNBC-specific miRNAs viz., miR153-1, miR4767, miR4487, miR6720, and miR-LET7I were exclusively found in both the TNBC cell lines. Identification of target genes belonging to TNBC-specific miRNAs was carried out (Supplementary Tables 7, 8). It is to be mentioned that with the cutoff criteria for target-gene identification used in this study (refer to Methods section), no targets were found for miR153-1. Of the five miRNA promoters found to be upregulated in the TNBC cell lines, 3 miRNAs, viz., miR-153-1, miR-6720, and miR-LET7I were found to have similar relative expression in TCGA data samples. Of these, miR-LET7I was found to have higher expression in TNBC (Supplementary Fig. 6).
Eight miRNAs obtained are found to be common across two cancer subtypes (Table 4). miR4512 was observed in all cancerous cell lines, both luminal-A (MCF7 and ZR751) and TNBC (MB231 and MB436) subtypes. miR3180-3 was observed in luminal-A (MCF7 and ZR751) and TNBC subtypes (MB231 and MB436). miR6791 and miR330 were observed to be common in three cancer cell lines, two luminal-A (MCF7 and ZR751) and one TNBC (MB231) subtypes. miR5787, miR6733, and miR3613 were observed in three cancer cell lines, two TNBC (MB231 and MB436) and luminal-A (ZR751) subtypes. miR6080 was observed to be present in three cancer cell lines, two TNBC (MB231 and MB436) and luminal-A (MCF7) subtypes. All the eight miRNAs listed above have been used for further downstream analysis to identify their putative gene targets based on mRNA expression data. Of the eight miRNA promoters found to be upregulated across breast cancer cell lines, the relative expression of three miRNAs, viz., miR-330, miR-3613, and miR-6733 were found to be complementary in studies reported in TCGA data samples using UALCAN webserver (Supplementary Fig. 7). The relative expression of the other five miRNAs in this resource was found to be insufficient to draw any conclusion.
All the RNA-seq datasets passed the quality check (>28) and hence were retained for further analyses (Supplementary Fig. 8). About 96% reads mapped for cell lines MCF10A, MCF7, and MB231 whereas, for cell lines ZR751 and MB436 >93% mapping was observed (Supplementary Table 9). In normal-like vs. luminal-A type, a total of 1,189 genes were upregulated (Supplementary Table 10) and 687 genes were downregulated (Supplementary Table 11). In normal-like vs. TNBC type, a total of 954 genes were upregulated (Supplementary Table 12) and 167 genes were downregulated (Supplementary Tables 13, 14, Supplementary Fig. 9). Five miRNAs specific to the TNBC cell lines were further studied to identify their binding to downregulated 3ŌĆ▓-UTR gene targets (Supplementary Tables 8, 15).
TNBC-specific miRNA target analysis of the downregulated genes helped in the identification of 13 genes (Supplementary Table 7). Of the 13 genes, ADAMTSL1, STC2, CPA4, and NUPR1 have been previously reported (Supplementary Table 6). It is interesting to note that FOXL2 is a target for multiple miRNAs, viz., miR4767, miR4487, and miR6720 in TNBC cell lines. Comparison of these target genes to other breast cancer cell lines from the CCLE database revealed that all of them have low expression as compared to the ACTB control gene (Supplementary Fig. 10). Genes STC2, CPA4, and NUPR1 were found to have a relatively higher expression amongst the 13 target genes of TNBC cell lines. In larger breast cancer samples obtained from TCGA, with the exception of SPOCK2, CPA4, C1orf228, and NFE2, the other target genes are found to have relatively low expression in TNBC as compared to normal samples (Supplementary Fig. 11). Relative expression of these genes in other cancer subtypes hints at the down-regulatory effect of TNBC-specific miRNAs. Survival plots of most of the downregulated genes (with the exception of NUPR1, CPA4, EPHA3, ADAMTSL1, and ATP13A4) were found to be associated with poor patient survival (Supplementary Fig. 12).
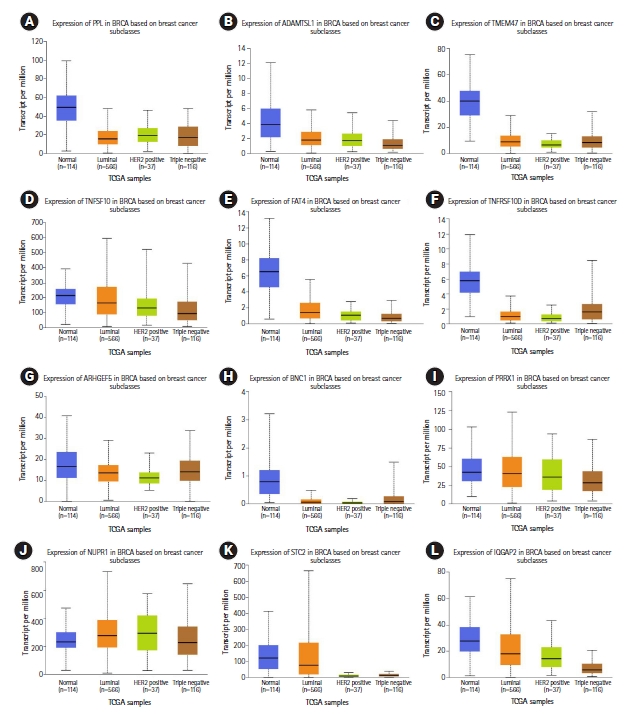
Eight miRNA promoters that are common across more than three cancer cell lines were also used as probes to identify the gene targets (Table 4, Supplementary Tables 16ŌĆÆ18). A total of 44 downregulated gene targets were identified across luminal-A and TNBC subtypes. In normal-like (MCF10A) vs. luminal-A (MCF7 and ZR751) downregulated genes, 17 genes have been predicted and their role in breast cancer has been reported earlier (RERG, IGFBP6, SPATA18, AXL, BMF, FXYD5, PTRF, RUNX2, UGT8, CFB, CSF3, HEG1, PLAU, PTER, S100A3, SNURF, and WIPF1) [35-51] (Fig. 4, Supplementary Tables 6, 19, Supplementary Fig. 13). In normal-like (MCF10A) vs. TNBC (MB231 and MB436) downregulated genes, 15 genes have been predicted in this study and their role in breast cancer have also been previously reported (TNFSF10, TMEM47, IQGAP2, FAT4, NUPR1, HOXC13, PRRX1, STC2, AC108941.2, ADAMTSL1, ARHGEF5, BNC1, CPA4, PPL, and TNFRSF10D) [52-66] (Fig. 5, Supplementary Tables 6, 20, Supplementary Fig. 14).
Of the remaining 12 target genes identified, nine genes in luminal-A were identified to be regulated by their corresponding miRNAs (gene A4GALT targeted by miR3180-3, miR4512, and miR6791; gene C10orf55 targeted by miR330, miR3180-3, miR5787, and miR6791; gene C2orf74 targeted by miR330 and miR5787; gene ZC4H2 targeted by miR330 and miR5787; gene ZNF512 targeted by miR330, miR3180-3, miR5787, and miR6791; gene ZNF655 targeted by miR5787; gene ZNF71 targeted by miR5787 and miR6791; gene HCG2042738 targeted by miR6791; gene HRCT1 targeted by miR4512 and miR5787) (Table 5). Similarly, three genes in TNBC were also identified to be regulated by their corresponding miRNAs (gene HIST3H2A targeted by miR6791; ZNF608 targeted by miR5787; ELOVL4 targeted by miR5787) (Supplementary Table 18). Comparison of these 12 target genes to other breast cancer cell lines from the CCLE database revealed that all of them have low expression as compared to the ACTB control gene (Supplementary Fig. 15). Genes HIST3H2A and C2ORF74 were found to have a relatively higher expression amongst the 12 target genes. In larger datasets of breast cancer, with the exception of ZNF71 and HIST3H2A, all other gene targets were found to be downregulated (Supplementary Figs. 13, 14, 16ŌĆÆ18). This observation supports the probable role of miRNA-mRNA axis in gene regulation. The down-regulation of A4GALT, C2ORF74, HRCT1, ZC4H2, ZNF512, ZNF655, ZNF608, and HIST3H2A genes were found to be independently associated with poor survival in breast cancer patients (Table 5, Supplementary Fig. 19). It needs to be mentioned that relative expression data and survival plots for gene HCG2042738 could not be obtained due to insufficient annotation.
The interplay between epigenetic gene regulation through histone modifications and other regulatory mechanisms like ncRNA is of great interest in cancer biology. In the present analysis, the role of H3K4me3 in miRNA expression based on promoter level peaks has been studied using ChIP-Seq and RNA-seq data integration. To achieve the same, a novel approach of mapping data derived from ChIP-Seq (miRNA promoter peaks) and RNA-Seq (targets of 3ŌĆ▓-UTRs of genes binding to miRNA) was used to understand epigenetic regulation that may aid in the identification of subtype and cell line specific miRNAs [15,16].
In normal-like cell line MCF10A, of the nine unique miRNAs identified, miR4530 was found to have a role in the suppression of cell proliferation, promote angiogenesis and induce apoptosis by targeting gene VASH1 (Vasohibin 1) in breast carcinoma [67]. Hence, promoter-level epigenetic regulation of miR4530 by H3K4me3 may have a protective role in normal-like subtypes. miR34B was observed to be present only in cell line MB436 (TNBC subtype). miR34B has high expression in TNBC tumors compared to normal types. Expression of miR34B highly correlates with clinical outcome of patients. Notch2 (notch receptor 2) gene that has a role controlling cell differentiation, is a direct target for miR34B [68]. miR6875 was observed in TNBC cell line MB436. According to previous reports, a high expression of miR6875 was observed in early breast cancer patients [69]. miR574-5p attenuates proliferation, migration, and epithelial mesenchymal transition (EMT) in TNBC cells by targeting genes BCL11A (BAF chromatin remodeling complex subunit) and SOX2 (SRY-Box transcription factor 2) to inhibit the SKIL (SKI like proto-oncogene)/TAZ (Tafazzin)/CTGF (connective tissue growth factor) axis [70].
Of the five TNBC subtypeŌĆÆspecific miRNAs, mir153, miR6720, and miR-LET7I were found to be upregulated in larger breast cancer datasets belonging to TCGA. miR153 has been reported to have a tumor suppressor role and has been suggested as a prognostic marker for TNBC [34].
The majority of the predicted gene targets (total 44) overlap with previous experimental studies and include 32 gene targets (Figs. 4,5) of eight miRNAs (miR4512, miR6791, miR330, miR3180-3, miR6080, miR5787, miR6733, and miR3613) which are identified in more than three breast cancer cell lines and absent in normal-like cell lines. Overexpression of miR330-3p in breast cancer cell lines has been reported earlier, which results in greater invasiveness in-vitro, and miR330-3p-overexpressing cells also metastasize more aggressively ex-ovo [71]. Gene CCBE1 (collagen and calcium binding EGF domains 1) is a direct target of miR330-3p, and knockout of CCBE1 results in a greater invasive capacity [71]. Exosomal expression of miR3613-3p promotes breast cancer cell proliferation and metastasis. It has been previously reported that miR3613-3p levels were negatively correlated to SOCS2 (suppressor of cytokine signaling 2) expression in breast cancer tissues [72]. Few genes were observed to be targeted by multiple miRNAs (like A4GALT and FOXL2 targeted by three miRNAs each) as it is known that miRNAs can regulate multiple targets based on seed match and sequence similarity between miRNA-mRNA [10].
Of the remaining 12 gene targets, relative gene expression of genes A4GALT, C2ORF74, HRCT1, ZC4H2, ZNF512, ZNF655, and ZNF608 agree with the proposed hypothesis of H3K4me3 regulated miRNA-mRNA axis in large patient data (TCGA samples) along with their relative expression in other breast cancer cell lines (CCLE database). These genes were associated with poor survival based on KM plots (Human Protein Atlas). The proposed methodology of miRNA-mRNA regulation when analyzed in the context of other histone modifications like H3K27me3, H3K4me1, H3K9me3 will enable better insights into the underlying mechanism of breast cancer regulation.
Notes
Acknowledgments
The authors thank the BRAF facility of C-DAC for HPC infrastructure. The authors would also like to deeply thank Dr. Janaki C.H. for her scientific review and critical comments. This research work is funded by the National Supercomputing Mission (NSM) of the Government of India. The authors thank anonymous reviewers for their valuable suggestions and constructive criticism in improving the manuscriptŌĆÖ before the funding statement.
Supplementary Materials
Supplementary data can be found with this article online at http://www.genominfo.org.
Supplementary┬ĀTable┬Ā1.
Gene Expression Omnibus (GEO) accession numbers for H3K4me3 chromatin immunoprecipitation sequencing data
Supplementary┬ĀTable┬Ā2.
Gene Expression Omnibus (GEO) accession numbers for input (control) chromatin immunoprecipitation sequencing data
Supplementary┬ĀTable┬Ā3.
Gene Expression Omnibus (GEO) accession numbers for RNA-sequencing data pertaining to breast cancer cell lines
Supplementary┬ĀTable┬Ā4.
Identification of significant threshold for peaks predicted using the IDR tool based on pseudoreplicates
Supplementary┬ĀTable┬Ā6.
RNA hybrid results of predicted differentially expressed miRNAs observed in more than three cancer cell lines with binding energy Ōēż ŌĆÆ25 kcal/mol. Genes that have been validated previously for their role in breast cancer (BC) are represented with underline. Genes that are highlighted in bold represent no previous literature support in BC.
Supplementary┬ĀTable┬Ā7.
Target gene list of five miRNAs present exclusively in TNBC subtype
Supplementary┬ĀTable┬Ā8.
RNA hybrid analysis of triple-negative breast cancer exclusive miRNA target genes
Supplementary┬ĀTable┬Ā10.
List of genes upregulated in MCF10A vs. luminal-A cell lines
Supplementary┬ĀTable┬Ā11.
List of genes downregulated in normal-like vs. luminal-A cell lines
Supplementary┬ĀTable┬Ā12.
List of genes upregulated in MCF10A vs. triple-negative breast cancer cell lines
Supplementary┬ĀTable┬Ā13.
Differentially expressed genes in normal-like and cancer cell lines
Supplementary┬ĀTable┬Ā14.
List of genes downregulated in normal-like vs. triple-negative breast cancer cell lines
Supplementary┬ĀTable┬Ā15.
List of five miRNA sequences (exclusively present in triple-negative breast cancer subtype) used for target-gene identification
Supplementary┬ĀTable┬Ā16.
List of eight miRNA sequences present in at least three breast cell lines used for target-gene identification
Supplementary┬ĀTable┬Ā17.
RNA hybrid analysis of miRNAs (present in at least three breast cancer cell lines) target genes (17 luminal-A and 15 triple-negative breast cancer) previously reported to have role in breast cancer
Supplementary┬ĀTable┬Ā18.
RNA hybrid analysis of miRNAs (present in at least three breast cancer cell lines) target genes with a probable role in breast cancer
Supplementary┬ĀTable┬Ā19.
Details of eight miRNAs and their 3'-untranslated region targets based on downregulated genes obtained from differential expression analysis of normal-like vs. luminal-A cell lines
Supplementary┬ĀTable┬Ā20.
Details of eight miRNAs and their 3'-untranslated region targets based on downregulated genes obtained from differential expression analysis of normal-like vs. triple-negative breast cancer cell lines
Supplementary┬ĀFig.┬Ā1.
Workflow for differential expression analysis using RNA-Seq data.
Supplementary┬ĀFig.┬Ā2.
FastQC output of chromatin immunoprecipitation sequencing data pertaining to each cell line and their corresponding biological replicates.
Supplementary┬ĀFig.┬Ā3.
Reproducibility analysis of replicates belonging to normal-like cell line (MCF10A).
Supplementary┬ĀFig.┬Ā4.
Reproducibility analysis of replicates belonging to luminal-A and triple-negative breast cancer cell lines. (A) Replicate 1 peak ranks versus Replicate 2 peak ranks - peaks that do not pass the threshold are colored red. (B) Replicate 1 log10 peak scores versus Replicate 2 log10 peak scores - peaks that do not pass the threshold are colored red. (C, D) Peak rank versus IDR scores are plotted in black.
Supplementary┬ĀFig.┬Ā5.
Annotation of peaks identified for each breast cancer cell line. ChIP, chromatin immunoprecipitation.
Supplementary┬ĀFig.┬Ā6.
Relative gene expression of triple-negative breast cancer subtype exclusive miRNAs from The Cancer Genome Atlas (TCGA) data samples. (A) miR-153-1. (B) miR-6720. (C) miR-Let7i.
Supplementary┬ĀFig.┬Ā7.
Relative gene expression of triple-negative breast cancer and luminal-A specific miRNAs from The Cancer Genome Atlas (TCGA) data samples. (A) miR-330. (B) miR-3613. (C) miR-6733.
Supplementary┬ĀFig.┬Ā8.
FastQC output of RNA-sequencing (RNA-Seq) data pertaining to each cell-line and their corresponding biological replicates.
Supplementary┬ĀFig.┬Ā9.
Differential expression analysis of RNA sequencing data: Volcano plots.
Supplementary┬ĀFig.┬Ā10.
Relative gene expression of triple-negative breast cancer subtype exclusive miRNA targets from the Broad Institute Cancer Cell Line Encyclopedia (CCLE) database.
Supplementary┬ĀFig.┬Ā11.
Relative gene expression of triple-negative breast cancer subtype exclusive miRNA targets from The Cancer Genome Atlas (TCGA) data samples.
Supplementary┬ĀFig.┬Ā12.
Five-year KM-survival plots from human protein atlas web server: triple-negative breast cancer subtype exclusive miRNA target genes.
Supplementary┬ĀFig.┬Ā13.
Relative gene expression of (The Cancer Genome Atlas [TCGA] breast cancer samples) luminal-A downregulated gene targets (5 of the total 17 genes) previously reported in breast cancer that do not correlate with predicted miRNA binding analysis.
Supplementary┬ĀFig.┬Ā14.
Relative gene expression of (The Cancer Genome Atlas [TCGA] breast cancer samples) triple-negative breast cancer downregulated gene targets (2 out of total 15 genes) previously reported in breast cancer predicted that do not correlate with predicted miRNA binding analysis.
Supplementary┬ĀFig.┬Ā15.
Relative gene expression of triple-negative breast cancer and luminal-A specific miRNAs gene targets from Broad Institute Cancer Cell Line Encyclopedia (CCLE) database (ACTB gene expression as control).
Supplementary┬ĀFig.┬Ā16.
Relative gene expression of triple-negative breast cancer and luminal-A specific miRNAs gene targets in The Cancer Genome Atlas (TCGA) samples: stage-wise expression.
Supplementary┬ĀFig.┬Ā17.
Relative gene expression of triple-negative breast cancer and luminal-A specific miRNAs gene targets in The Cancer Genome Atlas (TCGA) samples.
Supplementary┬ĀFig.┬Ā18.
Relative gene expression of triple-negative breast cancer and luminal-A specific miRNAs gene targets in The Cancer Genome Atlas (TCGA) samples:across cancer subtypes.
Supplementary┬ĀFig.┬Ā19.
Five-year KM-survival plots from Human Protein Atlas: triple-negative breast cancer and luminal-A specific miRNAs gene targets.
Fig.┬Ā1.
(A) Chromatin immunoprecipitation sequencing (ChIP-Seq) peak calling workflow for miRNA promoter prediction. (B) ChIP-Seq and RNA sequencing (RNA-Seq) data integration workflow for prediction of miRNA-mRNA interaction via 3ŌĆ▓-untranslated region (3ŌĆ▓-UTR) binding target prediction.
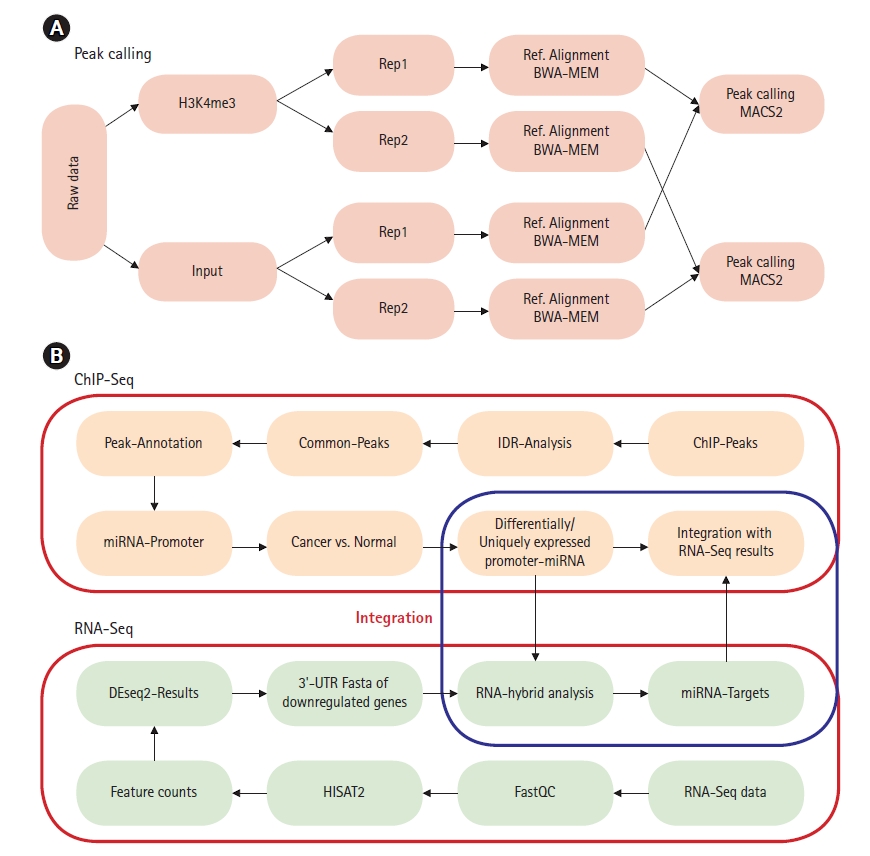
Fig.┬Ā2.
Total number of peaks predicted using MACS2 for biological replicates with H3K4me3 histone modification (p = 0.001). Replicate 1 and 2 are coloured as green and blue respectively.

Fig.┬Ā4.
Relative gene expression (The Cancer Genome Atlas [TCGA] breast cancer samples) of luminal-A downregulated gene targets (12 of the total 17 genes) previously reported in breast cancer that correlate with predicted miRNA binding analysis: (A) PTER, (B) HEG1, (C) SPATA18, (D) PTRF, (E) SNURF, (F) RERG, (G) AXL, (H) FXYD5, (I) WIPF1, (J) CSF3, (K) UGT8, and (L) IGFBP6.

Fig.┬Ā5.
Relative gene expression (TCGA breast cancer samples) of triple-negative breast cancer downregulated gene targets (12 of the total 15 genes) previously reported in breast cancer that correlate with predicted miRNA binding analysis: (A) PPL, (B) ADAMTSL1, (C) TMEM47, (D) TNFSF10, (E) FAT4, (F) TNFRSF10D, (G) ARHGEF5, (H) BNC1, (I) PRRX1, (J) NUPR1, (K) STC2, and (L) IQGAP2.
Table┬Ā1.
Details of overlapping peaks obtained after IDR analysis of all cell lines (biological replicates)
Table┬Ā2.
List of H3K4me3 regulated miRNA promoter-specific peaks across cell lines
| Subtype | Cell line | No. of peaks |
|---|---|---|
| Normal | MCF10A | 53 |
| Luminal-A | MCF7 | 44 |
| ZR751 | 44 | |
| TNBC | MB231 | 42 |
| MB436 | 54 |
Table┬Ā3.
Predicted cell line specific miRNAs
Table┬Ā4.
Predicted TNBC and luminal-A specific miRNAs common across (Ōēź3) cancer cell lines
Table┬Ā5.
Predicted gene targets of differentially regulated miRNAs in breast cancer cell lines (TNBC and luminal-A) proposed using ChIP-SeqŌĆÆRNA-Seq integrated analysis
References
2. Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 2006;295:2492ŌĆō2502.


3. Collignon J, Lousberg L, Schroeder H, Jerusalem G. Triple-negative breast cancer: treatment challenges and solutions. Breast Cancer (Dove Med Press) 2016;8:93ŌĆō107.


4. Luan QX, Zhang BG, Li XJ, Guo MY. MiR-129-5p is downregulated in breast cancer cells partly due to promoter H3K27m3 modification and regulates epithelial-mesenchymal transition and multi-drug resistance. Eur Rev Med Pharmacol Sci 2016;20:4257ŌĆō4265.

5. Yan H, Tian S, Slager SL, Sun Z. ChIP-seq in studying epigenetic mechanisms of disease and promoting precision medicine: progresses and future directions. Epigenomics 2016;8:1239ŌĆō1258.


6. Furey TS. ChIP-seq and beyond: new and improved methodologies to detect and characterize protein-DNA interactions. Nat Rev Genet 2012;13:840ŌĆō852.



8. Zhou VW, Goren A, Bernstein BE. Charting histone modifications and the functional organization of mammalian genomes. Nat Rev Genet 2011;12:7ŌĆō18.


9. Prabhu KS, Raza A, Karedath T, Raza SS, Fathima H, Ahmed EI, et al. Non-coding RNAs as regulators and markers for targeting of breast cancer and cancer stem cells. Cancers (Basel) 2020;12:351.



11. Place RF, Li LC, Pookot D, Noonan EJ, Dahiya R. MicroRNA-373 induces expression of genes with complementary promoter sequences. Proc Natl Acad Sci U S A 2008;105:1608ŌĆō1613.



12. Brennecke J, Stark A, Russell RB, Cohen SM. Principles of microRNA-target recognition. PLoS Biol 2005;3:e85.



13. Iorio MV, Ferracin M, Liu CG, Veronese A, Spizzo R, Sabbioni S, et al. MicroRNA gene expression deregulation in human breast cancer. Cancer Res 2005;65:7065ŌĆō7070.


14. Khalife H, Skafi N, Fayyad-Kazan M, Badran B. MicroRNAs in breast cancer: new maestros defining the melody. Cancer Genet 2020;246-247:18ŌĆō40.


15. Ramos J, Felty Q, Roy D. Integrated Chip-Seq and RNA-Seq data analysis coupled with bioinformatics approaches to investigate regulatory landscape of transcription modulators in breast cancer cells. Methods Mol Biol 2020;2102:35ŌĆō59.


16. Franco HL, Nagari A, Malladi VS, Li W, Xi Y, Richardson D, et al. Enhancer transcription reveals subtype-specific gene expression programs controlling breast cancer pathogenesis. Genome Res 2018;28:159ŌĆō170.



17. Andrews S. FASTQC: a quality control tool for high throughput sequence data. Cambridge: Babraham Institute, 2010. Accessed 2021 May 30 Available from: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
18. Li H, Durbin R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 2010;26:589ŌĆō595.



19. Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009;25:2078ŌĆō2079.



20. Zhang Y, Liu T, Meyer CA, Eeckhoute J, Johnson DS, Bernstein BE, et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol 2008;9:R137.



21. Bailey T, Krajewski P, Ladunga I, Lefebvre C, Li Q, Liu T, et al. Practical guidelines for the comprehensive analysis of ChIP-seq data. PLoS Comput Biol 2013;9:e1003326.



22. Feng J, Liu T, Qin B, Zhang Y, Liu XS. Identifying ChIP-seq enrichment using MACS. Nat Protoc 2012;7:1728ŌĆō1740.


23. Li Q, Brown JB, Huang H, Bickel PJ. Measuring reproducibility of high-throughput experiments. Ann Appl Stat 2011;5:1752ŌĆō1779.

24. Landt SG, Marinov GK, Kundaje A, Kheradpour P, Pauli F, Batzoglou S, et al. ChIP-seq guidelines and practices of the ENCODE and modENCODE consortia. Genome Res 2012;22:1813ŌĆō1831.



25. Heinz S, Benner C, Spann N, Bertolino E, Lin YC, Laslo P, et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol Cell 2010;38:576ŌĆō589.



26. Shin H, Liu T, Manrai AK, Liu XS. CEAS: cis-regulatory element annotation system. Bioinformatics 2009;25:2605ŌĆō2606.


27. Kim D, Paggi JM, Park C, Bennett C, Salzberg SL. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 2019;37:907ŌĆō915.



28. Liao Y, Smyth GK, Shi W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014;30:923ŌĆō930.


29. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 2014;15:550.



30. Hollbacher B, Balazs K, Heinig M, Uhlenhaut NH. Seq-ing answers: current data integration approaches to uncover mechanisms of transcriptional regulation. Comput Struct Biotechnol J 2020;18:1330ŌĆō1341.



31. Rehmsmeier M, Steffen P, Hochsmann M, Giegerich R. Fast and effective prediction of microRNA/target duplexes. RNA 2004;10:1507ŌĆō1517.



32. Augustin R, Endres K, Reinhardt S, Kuhn PH, Lichtenthaler SF, Hansen J, et al. Computational identification and experimental validation of microRNAs binding to the Alzheimer-related gene ADAM10. BMC Med Genet 2012;13:35.



33. Chandrashekar DS, Bashel B, Balasubramanya SA, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia 2017;19:649ŌĆō658.



34. Shi D, Li Y, Fan L, Zhao Q, Tan B, Cui G. Upregulation of miR-153 inhibits triple-negative breast cancer progression by targeting ZEB2-mediated EMT and contributes to better prognosis. Onco Targets Ther 2019;12:9611ŌĆō9625.


35. Hsu PC, Ho JY, Yu CP. RERG involvement in the RAS pathway and ER-dependent transcription in breast cancer. J Clin Oncol 2019;37(15 Suppl):e14638.

36. Nikulin SV, Raigorodskaya MP, Poloznikov AA, Zakharova GS, Schumacher U, Wicklein D, et al. In vitro model for studying of the role of IGFBP6 gene in breast cancer metastasizing. Bull Exp Biol Med 2018;164:688ŌĆō692.


37. Bornstein C, Brosh R, Molchadsky A, Madar S, Kogan-Sakin I, Goldstein I, et al. SPATA18, a spermatogenesis-associated gene, is a novel transcriptional target of p53 and p63. Mol Cell Biol 2011;31:1679ŌĆō1689.



39. Hornsveld M, Tenhagen M, van de Ven RA, Smits AM, van Triest MH, van Amersfoort M, et al. Restraining FOXO3-dependent transcriptional BMF activation underpins tumour growth and metastasis of E-cadherin-negative breast cancer. Cell Death Differ 2016;23:1483ŌĆō1492.



40. Lee YK, Lee SY, Park JR, Kim RJ, Kim SR, Roh KJ, et al. Dysadherin expression promotes the motility and survival of human breast cancer cells by AKT activation. Cancer Sci 2012;103:1280ŌĆō1289.



41. Bai L, Deng X, Li Q, Wang M, An W, Deli A, et al. Down-regulation of the cavin family proteins in breast cancer. J Cell Biochem 2012;113:322ŌĆō328.


42. McDonald L, Ferrari N, Terry A, Bell M, Mohammed ZM, Orange C, et al. RUNX2 correlates with subtype-specific breast cancer in a human tissue microarray, and ectopic expression of Runx2 perturbs differentiation in the mouse mammary gland. Dis Model Mech 2014;7:525ŌĆō534.


43. Cao Q, Chen X, Wu X, Liao R, Huang P, Tan Y, et al. Inhibition of UGT8 suppresses basal-like breast cancer progression by attenuating sulfatide-alphaVbeta5 axis. J Exp Med 2018;215:1679ŌĆō1692.



44. Suman S, Basak T, Gupta P, Mishra S, Kumar V, Sengupta S, et al. Quantitative proteomics revealed novel proteins associated with molecular subtypes of breast cancer. J Proteomics 2016;148:183ŌĆō193.


45. Hollmen M, Karaman S, Schwager S, Lisibach A, Christiansen AJ, Maksimow M, et al. G-CSF regulates macrophage phenotype and associates with poor overall survival in human triple-negative breast cancer. Oncoimmunology 2016;5:e1115177.


46. Zhao YR, Wang JL, Xu C, Li YM, Sun B, Yang LY. HEG1 indicates poor prognosis and promotes hepatocellular carcinoma invasion, metastasis, and EMT by activating Wnt/beta-catenin signaling. Clin Sci (Lond) 2019;133:1645ŌĆō1662.


47. Lin M, Zhang Z, Gao M, Yu H, Sheng H, Huang J. MicroRNA-193a-3p suppresses the colorectal cancer cell proliferation and progression through downregulating the PLAU expression. Cancer Manag Res 2019;11:5353ŌĆō5363.


48. Hung CM, Liu LC, Ho CT, Lin YC, Way TD. Pterostilbene enhances TRAIL-induced apoptosis through the induction of death receptors and downregulation of cell survival proteins in TRAIL-resistance triple negative breast cancer cells. J Agric Food Chem 2017;65:11179ŌĆō11191.


49. Gianni M, Terao M, Kurosaki M, Paroni G, Brunelli L, Pastorelli R, et al. S100A3 a partner protein regulating the stability/activity of RARalpha and PML-RARalpha in cellular models of breast/lung cancer and acute myeloid leukemia. Oncogene 2019;38:2482ŌĆō2500.


50. Saville B, Poukka H, Wormke M, Janne OA, Palvimo JJ, Stoner M, et al. Cooperative coactivation of estrogen receptor alpha in ZR-75 human breast cancer cells by SNURF and TATA-binding protein. J Biol Chem 2002;277:2485ŌĆō2497.


51. Garcia E, Machesky LM, Jones GE, Anton IM. WIP is necessary for matrix invasion by breast cancer cells. Eur J Cell Biol 2014;93:413ŌĆō423.


52. He W, Wang Q, Xu J, Xu X, Padilla MT, Ren G, et al. Attenuation of TNFSF10/TRAIL-induced apoptosis by an autophagic survival pathway involving TRAF2- and RIPK1/RIP1-mediated MAPK8/JNK activation. Autophagy 2012;8:1811ŌĆō1821.



53. Guo L, Zhang K, Bing Z. Application of a coexpression network for the analysis of aggressive and nonaggressive breast cancer cell lines to predict the clinical outcome of patients. Mol Med Rep 2017;16:7967ŌĆō7978.



54. Kumar D, Hassan MK, Pattanaik N, Mohapatra N, Dixit M. IQGAP2 acts as a tumor suppressor in breast cancer and its reduced expression promotes cancer growth and metastasis by MEK/ERK signalling pathways. Preprint at https://doi.org/10.1101/651034. 2019.

55. Hou L, Chen M, Zhao X, Li J, Deng S, Hu J, et al. FAT4 functions as a tumor suppressor in triple-negative breast cancer. Tumour Biol 2016;37:16337ŌĆō16343.

56. Chowdhury UR, Samant RS, Fodstad O, Shevde LA. Emerging role of nuclear protein 1 (NUPR1) in cancer biology. Cancer Metastasis Rev 2009;28:225ŌĆō232.


57. Li C, Cui J, Zou L, Zhu L, Wei W. Bioinformatics analysis of the expression of HOXC13 and its role in the prognosis of breast cancer. Oncol Lett 2020;19:899ŌĆō907.


58. Dong J, Lv Z, Chen Q, Wang X, Li F. PRRX1 drives tamoxifen therapy resistance through induction of epithelial-mesenchymal transition in MCF-7 breast cancer cells. Int J Clin Exp Pathol 2018;11:2629ŌĆō2635.


59. Hou J, Wang Z, Xu H, Yang L, Yu X, Yang Z, et al. Stanniocalicin 2 suppresses breast cancer cell migration and invasion via the PKC/claudin-1-mediated signaling. PLoS One 2015;10:e0122179.



60. Ji XW, Zhou TY, Lu Y, Wei MJ, Lu W, Cho WC. Breast cancer treatment and sulfotransferase. Expert Opin Ther Targets 2015;19:821ŌĆō834.


61. Li Z, Guo X, Wu Y, Li S, Yan J, Peng L, et al. Methylation profiling of 48 candidate genes in tumor and matched normal tissues from breast cancer patients. Breast Cancer Res Treat 2015;149:767ŌĆō779.


62. Debily MA, Camarca A, Ciullo M, Mayer C, El Marhomy S, Ba I, et al. Expression and molecular characterization of alternative transcripts of the ARHGEF5/TIM oncogene specific for human breast cancer. Hum Mol Genet 2004;13:323ŌĆō334.


63. Pangeni RP, Channathodiyil P, Huen DS, Eagles LW, Johal BK, Pasha D, et al. The GALNT9, BNC1 and CCDC8 genes are frequently epigenetically dysregulated in breast tumours that metastasise to the brain. Clin Epigenetics 2015;7:57.



64. Bademler S, Ucuncu MZ, Tilgen Vatansever C, Serilmez M, Ertin H, Karanlik H. Diagnostic and prognostic significance of carboxypeptidase A4 (CPA4) in breast cancer. Biomolecules 2019;9:103.



65. Sun C, Gu Y, Chen G, Du Y. Bioinformatics analysis of stromal molecular signatures associated with breast and prostate cancer. J Comput Biol 2019;26:1130ŌĆō1139.


66. Sanlioglu AD, Korcum AF, Pestereli E, Erdogan G, Karaveli S, Savas B, et al. TRAIL death receptor-4 expression positively correlates with the tumor grade in breast cancer patients with invasive ductal carcinoma. Int J Radiat Oncol Biol Phys 2007;69:716ŌĆō723.


67. Zhang T, Jing L, Li H, Ding L, Ai D, Lyu J, et al. MicroRNA-4530 promotes angiogenesis by targeting VASH1 in breast carcinoma cells. Oncol Lett 2017;14:111ŌĆō118.



68. Svoboda M, Sana J, Redova M, Navratil J, Palacova M, Fabian P, et al. MiR-34b is associated with clinical outcome in triple-negative breast cancer patients. Diagn Pathol 2012;7:31.



69. Xie Y, Du J, Liu Z, Zhang D, Yao X, Yang Y. MiR-6875-3p promotes the proliferation, invasion and metastasis of hepatocellular carcinoma via BTG2/FAK/Akt pathway. J Exp Clin Cancer Res 2019;38:7.



70. Zhang KJ, Hu Y, Luo N, Li X, Chen FY, Yuan JQ, et al. miR5745p attenuates proliferation, migration and EMT in triplenegative breast cancer cells by targeting BCL11A and SOX2 to inhibit the SKIL/TAZ/CTGF axis. Int J Oncol 2020;56:1240ŌĆō1251.


- TOOLS
-
-
 PDF Links
PDF Links
 PubReader
PubReader ePub Link
ePub Link XML Download
XML Download-
 Full text via DOI
Full text via DOI
 Full text via PMC
Full text via PMC-
 Download Citation
Download Citation
 Supplement1
Supplement1 Supplement2
Supplement2 Supplement3
Supplement3 Supplement4
Supplement4 Supplement5
Supplement5 Supplement6
Supplement6 Supplement7
Supplement7 Supplement8
Supplement8 Supplement9
Supplement9 Supplement10
Supplement10 Supplement11
Supplement11 Supplement12
Supplement12 Supplement13
Supplement13 Supplement14
Supplement14 Supplement15
Supplement15 Supplement16
Supplement16 Supplement17
Supplement17 Supplement18
Supplement18 Supplement19
Supplement19 Supplement20
Supplement20 Supplement21
Supplement21 Supplement22
Supplement22 Supplement23
Supplement23 Supplement24
Supplement24 Supplement25
Supplement25 Supplement26
Supplement26 Supplement27
Supplement27 Supplement28
Supplement28 Supplement29
Supplement29 Supplement30
Supplement30 Supplement31
Supplement31 Supplement32
Supplement32 Supplement33
Supplement33 Supplement34
Supplement34 Supplement35
Supplement35 Supplement36
Supplement36 Supplement37
Supplement37 Supplement38
Supplement38 Supplement39
Supplement39 Print
Print
-
-
METRICS

-
- 2 Crossref
- 0 Scopus
- 6,995 View
- 106 Download
- Related articles in GNI








