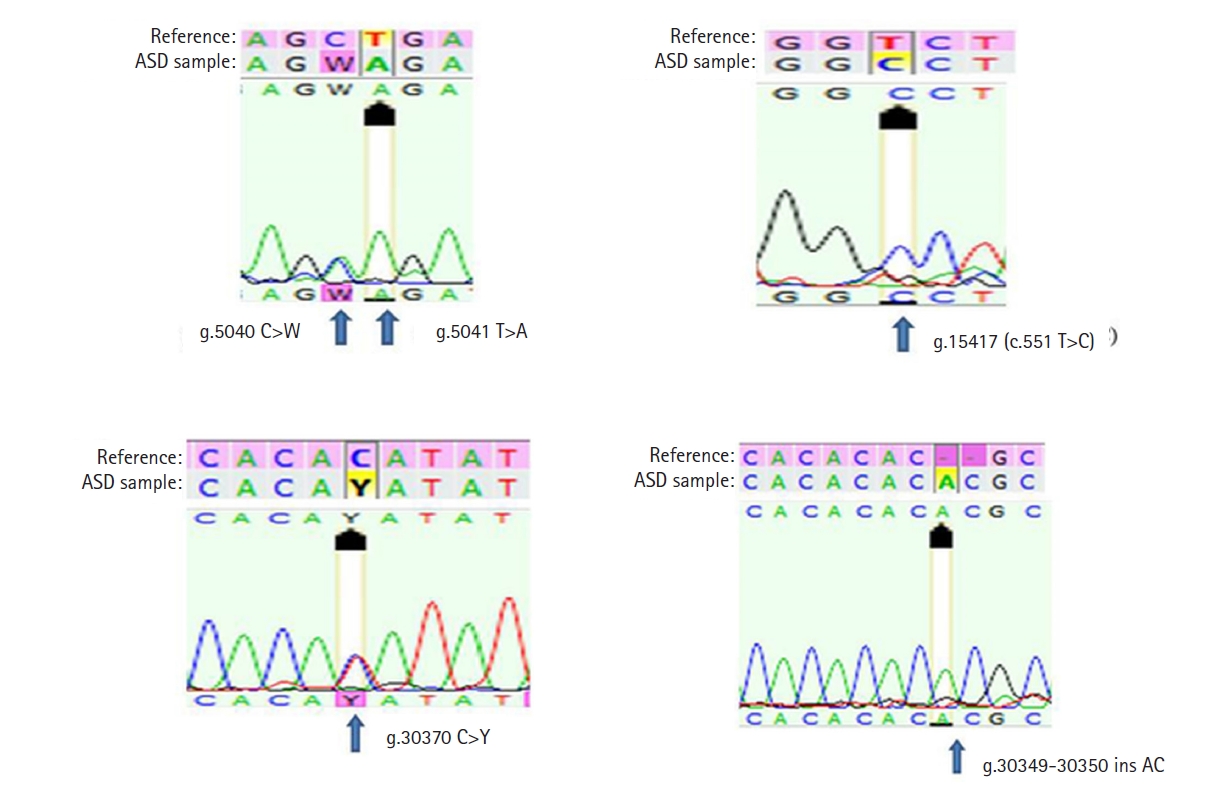
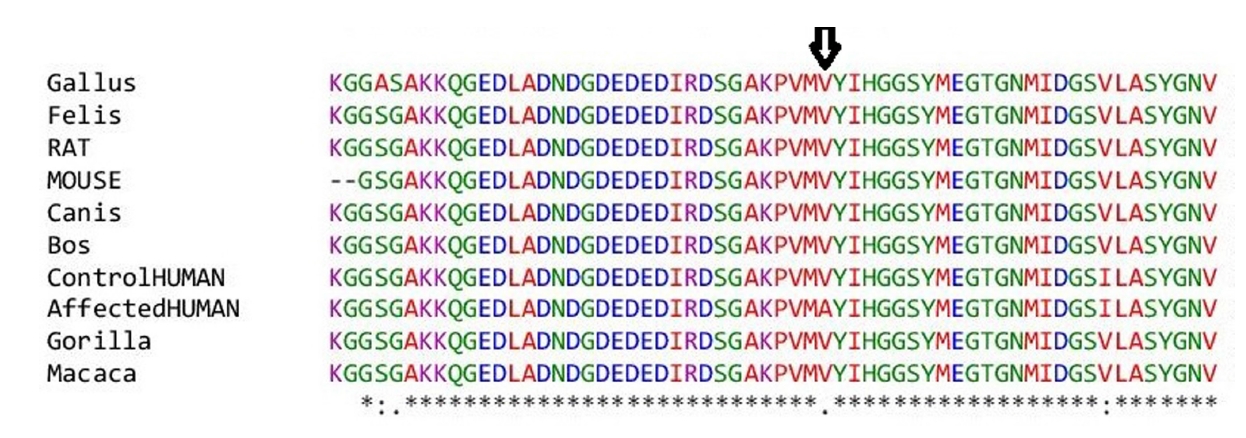
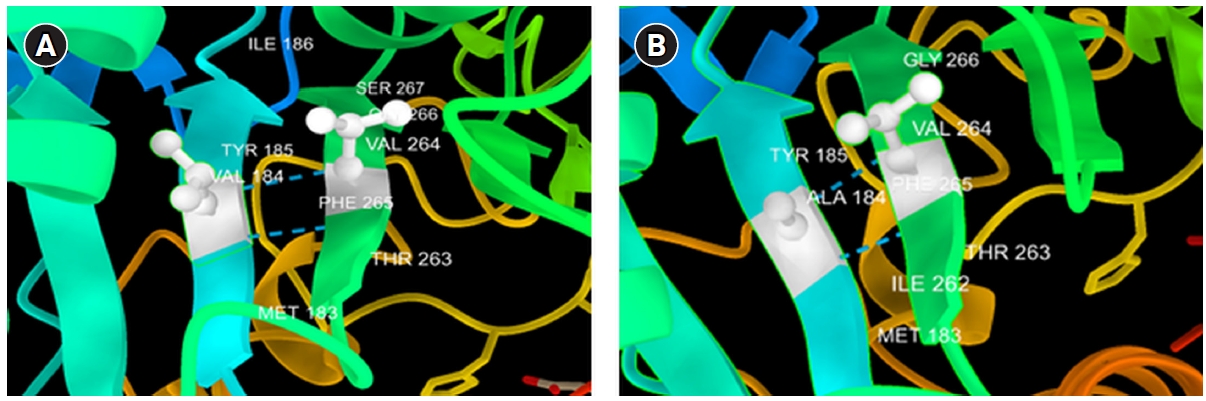
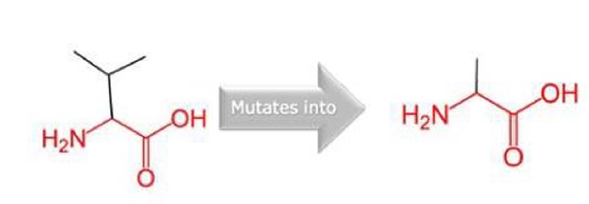
1. Spence SJ. The genetics of autism. Semin Pediatr Neurol 2004;11:196–204.
2. Chen JA, Penagarikano O, Belgard TG, Swarup V, Geschwind DH. The emerging picture of autism spectrum disorder: genetics and pathology. Annu Rev Pathol 2015;10:111–144.


3. Kanner L. Follow-up study of eleven autistic children originally reported in 1943. J Autism Child Schizophr 1971;1:119–145.


4. Pampanos A, Volaki K, Kanavakis E, Papandreou O, Youroukos S, Thomaidis L,
et al. A substitution involving the
NLGN4 gene associated with autistic behavior in the Greek population. Genet Test Mol Biomarkers 2009;13:611–615.


5. De Rubeis S, Buxbaum JD. Genetics and genomics of autism spectrum disorder: embracing complexity. Hum Mol Genet 2015;24:R24–R31.


7. Bourgeron T. Current knowledge on the genetics of autism and propositions for future research. C R Biol 2016;339:300–307.


8. de la Torre-Ubieta L, Won H, Stein JL, Geschwind DH. Advancing the understanding of autism disease mechanisms through genetics. Nat Med 2016;22:345–361.


9. Jamain S, Quach H, Betancur C, Rastam M, Colineaux C, Gillberg IC,
et al. Mutations of the X-linked genes encoding neuroligins
NLGN3 and
NLGN4 are associated with autism. Nat Genet 2003;34:27–29.



10. Raina SK, Chander V, Bhardwaj AK, Kumar D, Sharma S, Kashyap V,
et al. Prevalence of autism spectrum disorder among rural, urban, and tribal children (1-10 years of age). J Neurosci Rural Pract 2017;8:368–374.



12. Chen X, Liu H, Shim AH, Focia PJ, He X. Structural basis for synaptic adhesion mediated by neuroligin-neurexin interactions. Nat Struct Mol Biol 2008;15:50–56.


13. Nguyen TA, Lehr AW, Roche KW. Neuroligins and neurodevelopmental disorders: X-linked genetics. Front Synaptic Neurosci 2020;12:33.


14. Nguyen TA, Wu K, Pandey S, Lehr AW, Li Y, Bemben MA,
et al. A cluster of autism-associated variants on X-linked
NLGN4X functionally resemble
NLGN4Y. Neuron 2020;106:759–768.



15. Lawson-Yuen A, Saldivar JS, Sommer S, Picker J. Familial deletion within
NLGN4 associated with autism and Tourette syndrome. Eur J Hum Genet 2008;16:614–618.



16. Yan J, Oliveira G, Coutinho A, Yang C, Feng J, Katz C,
et al. Analysis of the neuroligin 3 and 4 genes in autism and other neuropsychiatric patients. Mol Psychiatry 2005;10:329–332.


17. Kent R, Simonoff E. Prevalence of anxiety in autism spectrum disorders. In: Anxiety in Children and Adolescents with Autism Spectrum Disorder: Evidence-Based Assessment and Treatment (Kerns CM, Renno P, Storch EA, Kendall PC, Wood JJ, eds.). San Diego: Elsevier Inc., 2017. pp. 5-32.
18. Volaki K, Pampanos A, Kitsiou-Tzeli S, Vrettou C, Oikonomakis V, Sofocleous C,
et al. Mutation screening in the Greek population and evaluation of
NLGN3 and
NLGN4X genes causal factors for autism. Psychiatr Genet 2013;23:198–203.


19. Ylisaukko-oja T, Rehnstrom K, Auranen M, Vanhala R, Alen R, Kempas E,
et al. Analysis of four neuroligin genes as candidates for autism. Eur J Hum Genet 2005;13:1285–1292.


20. Quartier A, Courraud J, Thi Ha T, McGillivray G, Isidor B, Rose K,
et al. Novel mutations in
NLGN3 causing autism spectrum disorder and cognitive impairment. Hum Mutat 2019;40:2021–2032.


21. Yu J, He X, Yao D, Li Z, Li H, Zhao Z. A sex-specific association of common variants of neuroligin genes (
NLGN3 and
NLGN4X) with autism spectrum disorders in a Chinese Han cohort. Behav Brain Funct 2011;7:13.



22. Redin C, Gerard B, Lauer J, Herenger Y, Muller J, Quartier A,
et al. Efficient strategy for the molecular diagnosis of intellectual disability using targeted high-throughput sequencing. J Med Genet 2014;51:724–736.


25. Blasi F, Bacchelli E, Pesaresi G, Carone S, Bailey AJ, Maestrini E,
et al. Absence of coding mutations in the X-linked genes neuroligin 3 and neuroligin 4 in individuals with autism from the IMGSAC collection. Am J Med Genet B Neuropsychiatr Genet 2006;141B:220–221.


26. Talebizadeh Z, Lam DY, Theodoro MF, Bittel DC, Lushington GH, Butler MG. Novel splice isoforms for
NLGN3 and
NLGN4 with possible implications in autism. J Med Genet 2006;43:e21.



27. Landini M, Merelli I, Raggi ME, Galluccio N, Ciceri F, Bonfanti A,
et al. Association analysis of noncoding variants in neuroligins 3 and 4X genes with autism spectrum disorder in an Italian cohort. Int J Mol Sci 2016;17:1765.